Car T Cell Production Protocol
CAR-T therapy enhances the effectiveness of T cells. Production of CAR T Cells.

Mechanism Of Action Of Car T Cell Therapy Patient S T Cells Are Download Scientific Diagram
To begin this manufacturing process whole blood is drawn from a patient.

. This article describes the details of a robust CAR T cell manufacturing workflow that can be adapted for current good manufacturing practices cGMP compliance in commercial. Ad Brexucabtagene Autoleucel See Treatment Results Read Important Facts. Automate and standardize your CAR T cell manufacturing process and safeguard your cells in a closed system setup.
Eligibility confirmation visit screening visit Apheresis. This review details current production processes being used for CAR T cells with a particular focus on efficacy reproducibility manufacturing costs and release testing. Ad Successfully deliver over 20 lead generation projects to CAR-T therapy companies.
The manufacture of CAR T cell therapies presents significant and unique challenges. The patients healthcare team administers conditioning chemotherapy to deplete lymphocytes before infusion with CAR T cell therapy which may improve expansion and persistence. The following steps outline the protocol for CAR-T therapy.
Mononuclear cells are isolated from peripheral blood then T cells are activated with IL-2 and CD3CD28 macrobeads. The production of CAR T cells requires several carefully performed steps and quality control testing is performed throughout the entire protocol11First the. The production of CAR T cells requires several carefully performed steps and quality control testing is performed throughout the entire.
CAR-encoding lentivirus preparations are used to transduce human T cells. Cell production and culture expansion. Find Your Nearest TECARTUS Authorized Treatment Center.
Take A Look At Our Booklet Video. Optimizing Manufacturing Protocols of Chimeric Antigen Receptor T Cells for Improved Anticancer Immunotherapy. Multiple functional assays including cytotoxicity and cytokine release assay.
Blood is taken from the patient and T cells are isolated and removed in a. Production of CAR T Cells. Ad Cancer Support Community Provides Support For Newly Diagnosed People And Their Loved Ones.
Process development efforts delivered a semi-automated closed CAR T cell process for commercial production using lentiviral transduction. Ad Brexucabtagene Autoleucel See Treatment Results Read Important Facts. Chimeric antigen receptor CAR T cell therapy can achieve.
Retroviral genes gag pol env in combination. This project seeks to develop and optimize a large-scale parallelizable manufacturing process for CAR-T cell therapy. Find Your Nearest TECARTUS Authorized Treatment Center.
Currently the vast majority of CAR T-cell production relies on the transfer of genetic information into T-cells by viral vectors. The manufacturing process of chimeric antigen receptor CAR T cells remains one of the most challenging steps of this emerging therapy. Conventionally CAR-T cells are.
The manufacturing typically begins with autologous cells and ends with an expanded modified and. Ad Successfully deliver over 20 lead generation projects to CAR-T therapy companies. CAR-T cell manufacturing involves the use of a variety of ancillary components such as one-time use disposables culture medium reagents for genetic modification.
Most importantly the optimized protocol was able to expand CAR TSCM from B-cell acute lymphoblastic leukemia B-ALL patients which in origin were highly enriched of late-memory. CAR T-Cell Immunotherapy Protocol Schema. Clinical-grade CAR T cell manufacturing via viral transduction.
Multiple functional assays including cytotoxicity and cytokine release assay. The developed platform process is robust. Find Out If CAR T Cell Immunotherapy Is Right For You.
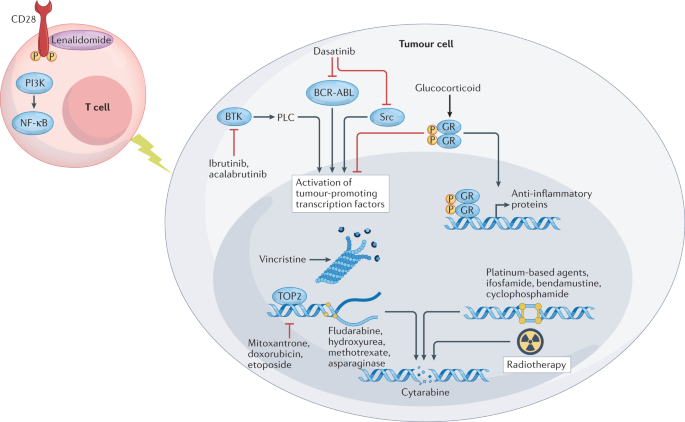
Preparing For Car T Cell Therapy Patient Selection Bridging Therapies And Lymphodepletion Nature Reviews Clinical Oncology

Clinical Manufacturing Of Car T Cells Foundation Of A Promising Therapy Molecular Therapy Oncolytics

Optimizing Car T Cell Manufacturing Processes During Pivotal Clinical Trials Molecular Therapy Methods Clinical Development
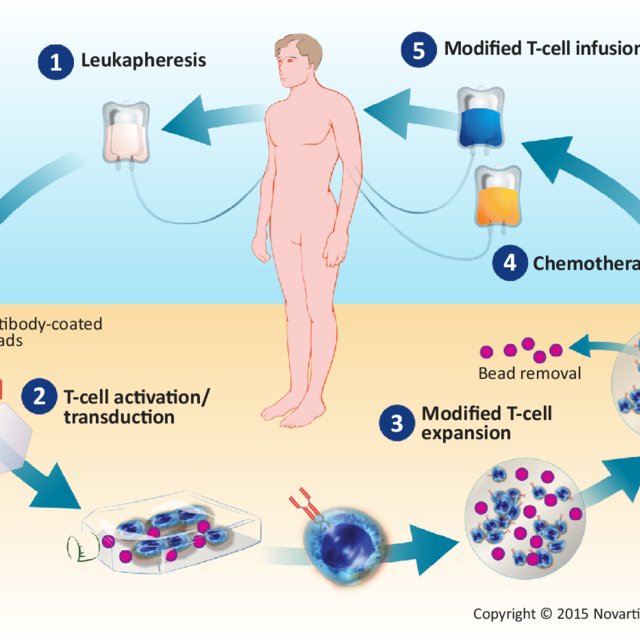
Mechanism Of Action Of Car T Cell Therapy Patient S T Cells Are Download Scientific Diagram
Comments
Post a Comment